Answer: Rust is a chemical reaction (oxidation) of iron that occurs over a period of time as the bare metal surface comes in contact with oxygen present in the air or water. Rust “eats” away the metal, rendering it weak and fragile. Avoid contact with water or moisture.
Why does rust happen?
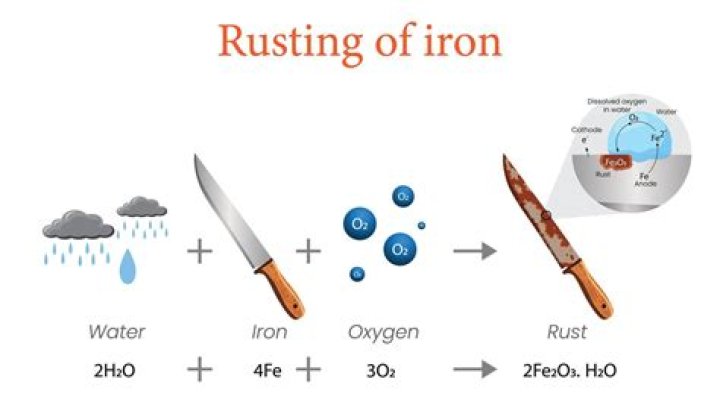
Rust results from a reaction called oxidation, in which iron reacts with water and oxygen to form hydrated iron (III) oxide. Essentially, the metal is naturally returning to its unrefined state. Learn how to remove rust with electrolysis. Because iron and oxygen have opposite charges, they gravitate toward each other.
What exactly is rust?
Most of us know rust as a reddish-brown flaky coat on metal and think nothing more of it, however rust is the term commonly used for the corrosion and oxidation of iron and its alloys, such as steel. Rust is very common, as iron reacts easily with oxygen. …
What is rust explain with example?
verb. Rust is a reddish-brown or reddish-yellow color that coats iron or steel when exposed to air and moisture. An example of rust is what happens to the chain on a bike that’s been left out in the rain all winter.
What is the formula of rust?
Rust is apparently a hydrated form of iron(III)oxide. The formula is approximately Fe2O3•32H2O, although the exact amount of water is variable.
How we can prevent rusting?
Here are some ways where we can prevent rusting :
- Paint it – Paint the metal with any durable acrylic paint.
- Keep moisture at Bay – Avoid contact with water or moisture.
- Coat it with Oil – Use of oil or grease significantly reduces the chances of developing rust in iron.
What kills rust?
For more stubborn rust, try using white vinegar. The acetic acid in this common household product is acidic enough to dissolve rust. You can soak smaller things like earrings, wipe it onto a surface with an old cloth, or just pour it directly over rust spots or bolts and screws that have rusted together.
What does rust do to your body?
Rust isn’t inherently harmful to human beings. In particular, touching rust or getting it on your skin isn’t associated with any health risks. While you can get tetanus from a wound caused by a rusty object, it’s not the rust that causes tetanus. Instead, it’s caused by a type of bacteria that may be on the object.
Is rust poisonous to humans?
What causes rust to form on a metal surface?
Because the air we breathe has moisture in it, oxidation will occur even if there is no water added to the metal. There is enough hydrogen and oxygen in the air to allow the atoms to bond with the iron. This creates the chemical reaction known as oxidation, or rust. There are things you can do to prevent rust from forming on your metal surfaces.
How does rusting occur in water and oxygen?
Rusting is an oxidation reaction. The iron reacts with water and oxygen to form hydrated iron (III) oxide, which we see as rust. Here is the word equation for the reaction: iron + water + oxygen → hydrated iron (III) oxide. Iron and steel rust when they come into contact with water and oxygen – both are needed for rusting to occur.
How does rust work as a chemical compound?
How does rust work? Rust is the common name for a very common compound, iron oxide. Iron oxide, the chemical Fe 2 O 3, is common because iron combines very readily with oxygen — so readily, in fact, that pure iron is only rarely found in nature.
How does rust form in a dry environment?
Left in a totally dry environment, iron or steel will not rust. It is when moisture is added that the oxidation process starts to occur. Because the air we breathe has moisture in it, oxidation will occur even if there is no water added to the metal.